We want to know how much heat energy is lost in this way. One is that the oxidative manufacture of methanol will be exothermic, so burning the methanol produced will yield less heat than would be produced by burning the methane from which it was produced. When we contemplate trying to develop a catalyst and a manufacturing plant to carry out this reaction, we soon discover reasons for wanting to know the enthalpy change. (At present, the economic feasibility of marine transport of liquefied natural gas, LNG, is marginal, but it appears to be improving.) This technology would make it possible to utilize the fuel value of known natural gas resources that are presently useless because they are located too far from population centers. 
However, the process involves two steps and requires a substantial capital investment.) If the cost of manufacturing methanol could be decreased sufficiently, it would become economically feasible to convert natural gas, which cannot be transported economically unless it is feasible to build a pipeline for the purpose, into liquid methanol, which is readily transported by ship. (Methanol is currently manufactured from methane. If the catalyst were not inordinately expensive or short-lived, and the operating pressure were sufficiently low, this would be an economical method for the manufacture of methanol. (This is frequently called a selective oxidation, to distinguish it from the non-selective oxidation that produces carbon dioxide and water.) Nevertheless, it would be very desirable to devise a catalyst that quantitatively-or nearly quantitatively-converted methane to methanol according to the equation In any circumstance, methanol is, at best, a trace product. If the reaction is done with an excess of methane, a portion of the carbon-containing product will be carbon monoxide rather than carbon dioxide. The normal products in the oxidation of methane are, of course, carbon dioxide and water. 
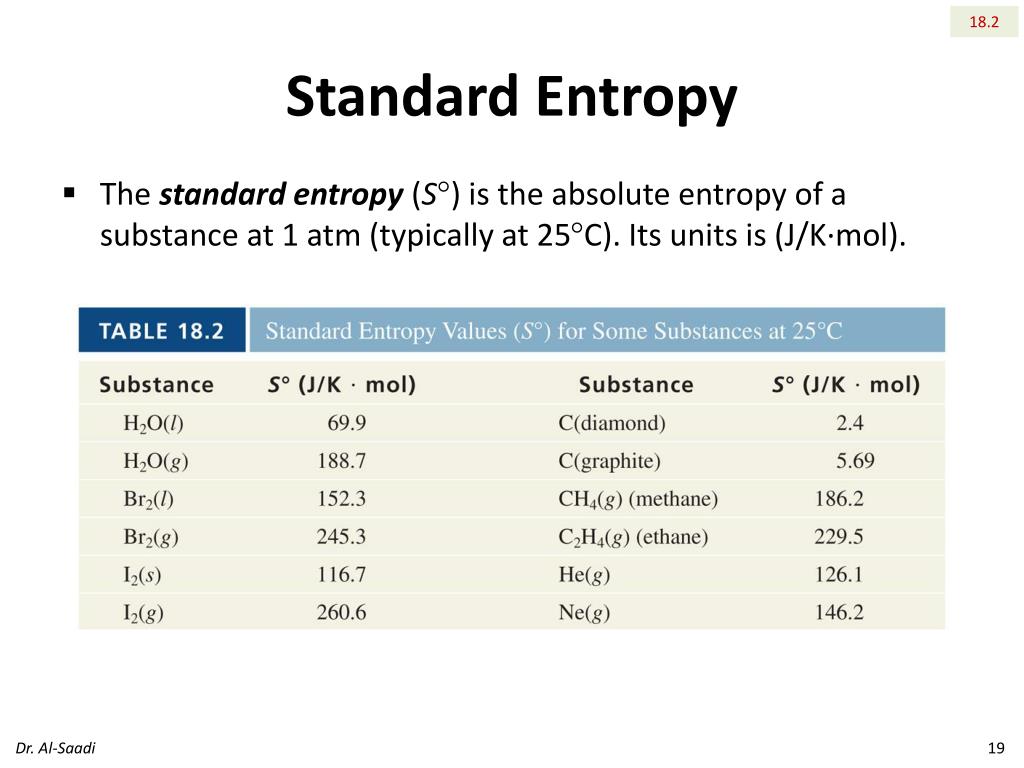
The oxidation of methane to methanol is a reaction that illustrates the value of this approach. (See below.) Because enthalpy is a state function, the enthalpy change that we calculate this way will be valid for any process that converts the specified reactants into the specified products. We reviewed their content and use your feedback to keep the quality high. Who are the experts Experts are tested by Chegg as specialists in their subject area. Then indicate which change (system or surroundings) must have a larger absolute value in each case and why. Question: Which species will have the lowest absolute entropy at 25☌ H2(g) SO3(e) CCl4 H2000) C6H1206(5) Show transcribed image text. When we multiply these chemical equations by the appropriately signed stoichiometric coefficient and add them, we obtain the chemical equation for the indicated reaction of A and B to give C and D. Predict and explain the sign of the entropy change for (i) the system, and (ii) the surroundings in each of the following processes. We can demonstrate this by writing out the chemical equations corresponding to the formation of A, B, C, and D from their elements. If we have the enthalpies of formation, we can compute the enthalpy change for the reaction. Under identical conditions, it is greater for a heavier gas.\nonumber \] Molar entropy is not the same for all gases. Entropy of gas at standard conditions (1 bar) Data from NIST Standard Reference Database 69: NIST Chemistry WebBook The National Institute of Standards and Technology (NIST) uses its best efforts to deliver a high quality copy of the Database and to verify that the data contained therein have been selected on the basis of sound scientific judgment. The standard molar entropy at pressure = P 0 These are often (but not necessarily) chosen to be the standard temperature and pressure. SO, since CF4 is the molecule with largest size and most complex (more particles such as protons, electrons, etc) it has the highest entropy of the molecules given. The more complex the molecule is, the higher the molar entropy. In chemistry, the standard molar entropy is the entropy content of one mole of pure substance at a standard state of pressure and any temperature of interest. Re: Determining Increasing Standard Molar Entropy. Standard entropy content of one mole of a substance under a standard state 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
